Aqualung Therapeutics Advances ALT-100 Program Targeting eNAMPT-TLR4 Signaling in Idiopathic Pulmonary Fibrosis
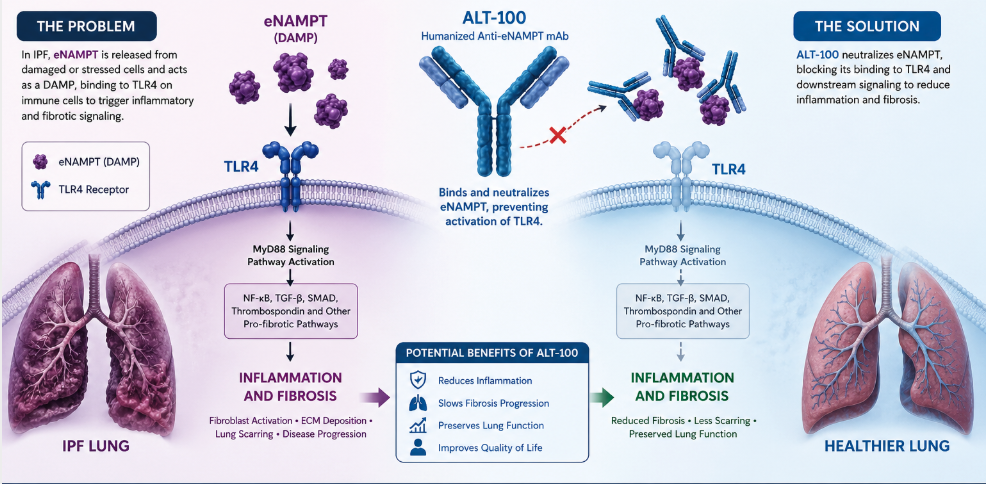
JUPITER, Fla. — Aqualung Therapeutics, in collaboration with the University of Florida, announced continued advancement of its lead therapeutic candidate, ALT-100, a humanized monoclonal antibody designed to neutralize extracellular NAMPT (eNAMPT) and disrupt TLR4-mediated inflammatory signaling in idiopathic pulmonary fibrosis (IPF).
IPF is a rapidly progressing and often fatal lung disease with limited treatment options. Currently approved therapies modestly slow disease progression but do not stop or reverse fibrosis. Many patients are not eligible for lung transplantation, leaving a significant unmet medical need for more effective interventions.
Aqualung’s approach targets eNAMPT, a damage-associated molecular pattern (DAMP) protein that activates Toll-like receptor 4 (TLR4), driving inflammatory and fibrotic pathways central to IPF progression. Elevated eNAMPT levels have been strongly associated with disease severity and reduced survival in IPF patients, positioning the pathway as a compelling therapeutic target.
ALT-100 has demonstrated the ability to attenuate fibrosis progression in multiple preclinical models, including radiation-induced and bleomycin-induced lung injury. The current research program focuses on optimizing subcutaneous dosing strategies to maximize therapeutic impact in established fibrosis. In parallel, studies are evaluating ALT-100 in combination with FDA-approved IPF therapies, including pirfenidone and nintedanib, to assess potential additive or synergistic effects.
“These studies are designed to generate a clear translational path from mechanism to patient impact,” said Stanley Miele, Project Leader. “By targeting a key upstream driver of inflammation and fibrosis, ALT-100 has the potential to meaningfully change how IPF is treated.”
Preclinical evaluations include comprehensive phenotyping across histologic, physiologic, and molecular endpoints, including lung stiffness, fibrosis scoring, key signaling proteins, and single-cell RNA sequencing to identify responsive cell populations and pathways.
The ALT-100 program is supported by encouraging clinical and preclinical safety data. Phase 1A studies in healthy volunteers reported no serious adverse events. The therapy is currently being evaluated in a Phase 2A clinical trial for acute respiratory distress syndrome (ARDS) (NCT05938036). Pharmacokinetic studies demonstrate a favorable half-life profile, with no observed toxicity in extended-duration animal studies.
This work reflects a strong academic and industry partnership between the University of Florida and Aqualung Therapeutics and is supported by NIH funding (Project Number: 1R41HL180035-01).
Successful completion of this program is expected to support future regulatory advancement and clinical development of ALT-100 as a novel therapeutic option for patients with IPF.
